Inflation Reduction Act (IRA) Signed into Law, Includes Multiple Medicare Drug Pricing Reforms
On Aug. 12, the Inflation Reduction Act of 2022 (IRA) passed the U.S. House by a vote of 220-207, and was signed into law by President Biden on August 16. First passed by the U.S. Senate on Aug. 7, the $740 billion budget reconciliation package includes policies on Medicare drug pricing, Affordable Care Act (ACA) subsidies, energy, climate, and taxes. This update provides high-level details on the notable health care-related provisions in the IRA.
Allowing Medicare to Negotiate Drug Prices
With the goal of improving affordability for high-priced drugs in Medicare Parts B and D, the IRA directs the Department of Health and Human Services (HHS) to establish a drug price negotiation program for certain high-priced, single-source drugs and biological products. Under this program, the HHS Secretary will publish a list of selected drugs that meet certain criteria, then negotiate (and renegotiate as needed) maximum fair prices with manufacturers of those drugs. Drugs eligible for negotiation include the 50 Part B and 50 Part D single-source drugs with the highest total expenditures during the most recent 12-month period; however, negotiation is limited to Part D drugs for 2026 and 2027. Negotiated prices must take effect for 10 eligible drugs in 2026, increasing to 20 drugs in 2029. For 2026, the expenditure period to be reviewed is June 1, 2022 through May 31, 2023, and the selected drug list publication date will be Sept. 1, 2023.
Redesigning the Medicare Part D Program, Including Capping Annual Out-of-Pocket Costs for Beneficiaries
The IRA significantly reforms the Medicare Part D benefit design, including capping maximum out-of-pocket (OOP) costs at $2,000 annually, with a copay smoothing component; capping annual premium growth at 6%; and expanding eligibility in the Low-Income Subsidy (LIS) program.
Beneficiary Cost-Sharing Changes:
- Beginning in 2024, beneficiaries will be responsible for $0 in the catastrophic benefit phase. There are no changes to the initial coverage phase or coverage gap phase.
- Beginning in 2025, the coverage gap phase will be eliminated, and a new $2,000 OOP cap will be applicable with the option to spread OOP payments out over the course of the year. The initial coverage phase remains unchanged.
Part D Benefit Design: The bill restructures plan, manufacturer, and federal government liabilities for the different benefit phases beginning in 2025:
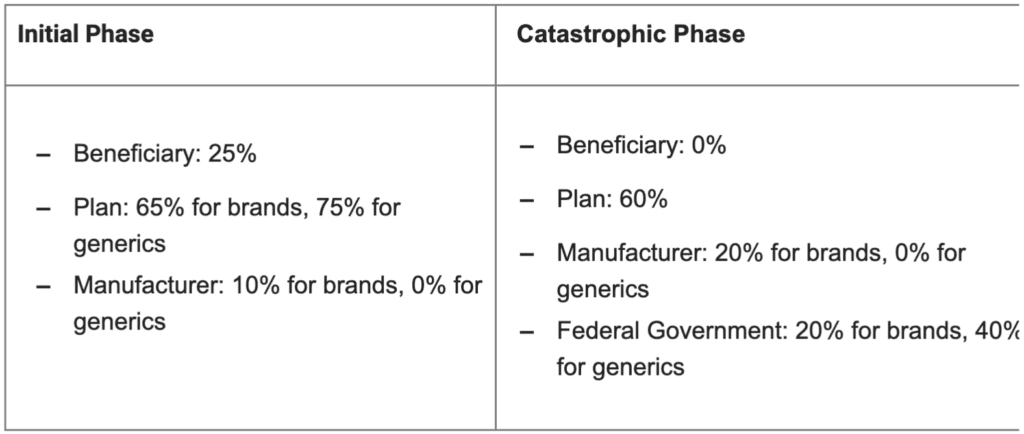
Premium Stabilization:
For 2024 through 2029, any increase in the Part D base beneficiary premium is limited to the lesser of a 6% increase from the previous year or the premium that would have been applied if the stabilization program was not established. In 2030 and subsequent years, the HHS Secretary is authorized to make adjustments necessary to the base Part D premium to ensure that premium is increased by the lesser of 6% or what the premium would have been if the stabilization program was not established.
•Expanded LIS Eligibility: The bill expands eligibility for the Part D LIS program from 135% of the federal poverty level to 150% beginning in 2024.
Capping Insulin Cost-Sharing in Medicare
For 2023 through 2025, the bill caps beneficiary cost-sharing at $35 a month for Medicare Part D or Medicare Advantage Prescription Drug Plan (MA-PD) covered insulin products. In 2026 and beyond, it caps cost-sharing at the lesser of $35 or 25% of the maximum fair price or 25% of the plan’s negotiated price. The cost-sharing is capped regardless of where the beneficiary is in the benefit phase, and Part D and MA-PD plans are eligible for a retroactive subsidy in 2023 equal to the aggregate reduction in cost-sharing and deductible due to implementing this provision.
Implementing Drug Manufacturer Inflationary Rebates in Medicare
The legislation requires drug manufacturers to pay rebates to the government if drug prices in Medicare Part B and Part D rise faster than inflation, with rebates equaling the rate at which the price of the drug exceeds inflation. This rebate provision goes into effect Jan. 1, 2023 for Part B rebatable drugs and Oct. 1, 2022 for Part D rebatable drugs. Drugs with an average cost of less than $100 are excluded. Additionally, HHS is instructed to reduce or waive the rebate amount for a Part D rebatable drug if it is on the drug shortage list, per the Federal Food, Drug, and Cosmetic Act.
Requiring Vaccine Coverage in Medicare Part D
Beginning in 2023, Part D plans are required to cover all adult vaccines recommended by the Advisory Committee on Immunization Practices, without cost-sharing or the application of a deductible (other than vaccines covered under Part B). Part D and MA-PD plans are eligible for a retroactive subsidy in 2023 equal to the aggregate reduction in cost-sharing and deductible due to implementing this provision.
Extended Delay of the Medicare Part D Rebate Rule
The legislation includes an additional five-year delay of the implementation of a rule that would prohibit manufacturer rebates in Part D, to Jan. 1, 2032.
Extending Enhanced ACA Subsidies Through 2025
Originally set to expire at the end of this year, the IRA extends the enhanced American Rescue Plan Act (ARPA) ACA premium tax credit subsidies through 2025.

